New Section on Risk Matrix Available Now in the ISN-ACT Toolkit
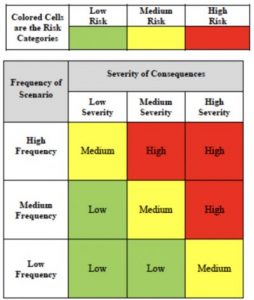
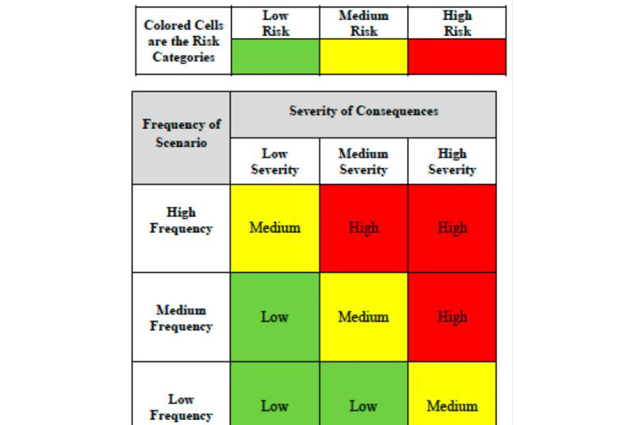
The ISN-ACT Toolkit has been extended to include a chapter on risk management – a fundamental pillar in the planning and executing of clinical trials that helps categorize risks based on their likelihood of occurrence and potential impact on the study.
This new section was contributed by ISN-Advancing Clinical Trials Committee member Dr. Paula Andrea Mariolo, a nephrologist at the Renálida Institution in Argentina. She and her colleagues developed a risk matrix to help improve risk management and maintain perspective during clinical trials.
Access the new content and learn how to use it here
Dr. Mariolo comments, “Implementing the risk matrix at Renálida has been crucial for improving risk management in our clinical trials. This tool has allowed us to identify, periodically review, and effectively mitigate potential risks, ensuring patient safety and the integrity of our studies.”
The ISN-ACT Toolkit provides an invaluable platform for sharing and promoting best practices in clinical research, fostering an environment of learning and continuous improvement.”
Explore more best practices in clinical trials in the ISN-ACT Clinical Trials Toolkit here.