Jan 2023 Edition

Impact Factor 2022
Recent publications in Kidney International Reports discuss the roles of glucose, blood pressure, and cardiovascular complications in chronic kidney disease.
Over the past three months, Kidney International has published several studies on the specific effects of SGLT2 inhibitors, lipotoxicity, genetics, endothelial stress responses, and environmental pollution on chronic kidney disease.
KIDNEY INTERNATIONAL ARTICLES |
Kidney Outcomes in Patients With Diabetes Mellitus Did Not Differ Between Individual Sodium-Glucose Cotransporter-2 Inhibitors
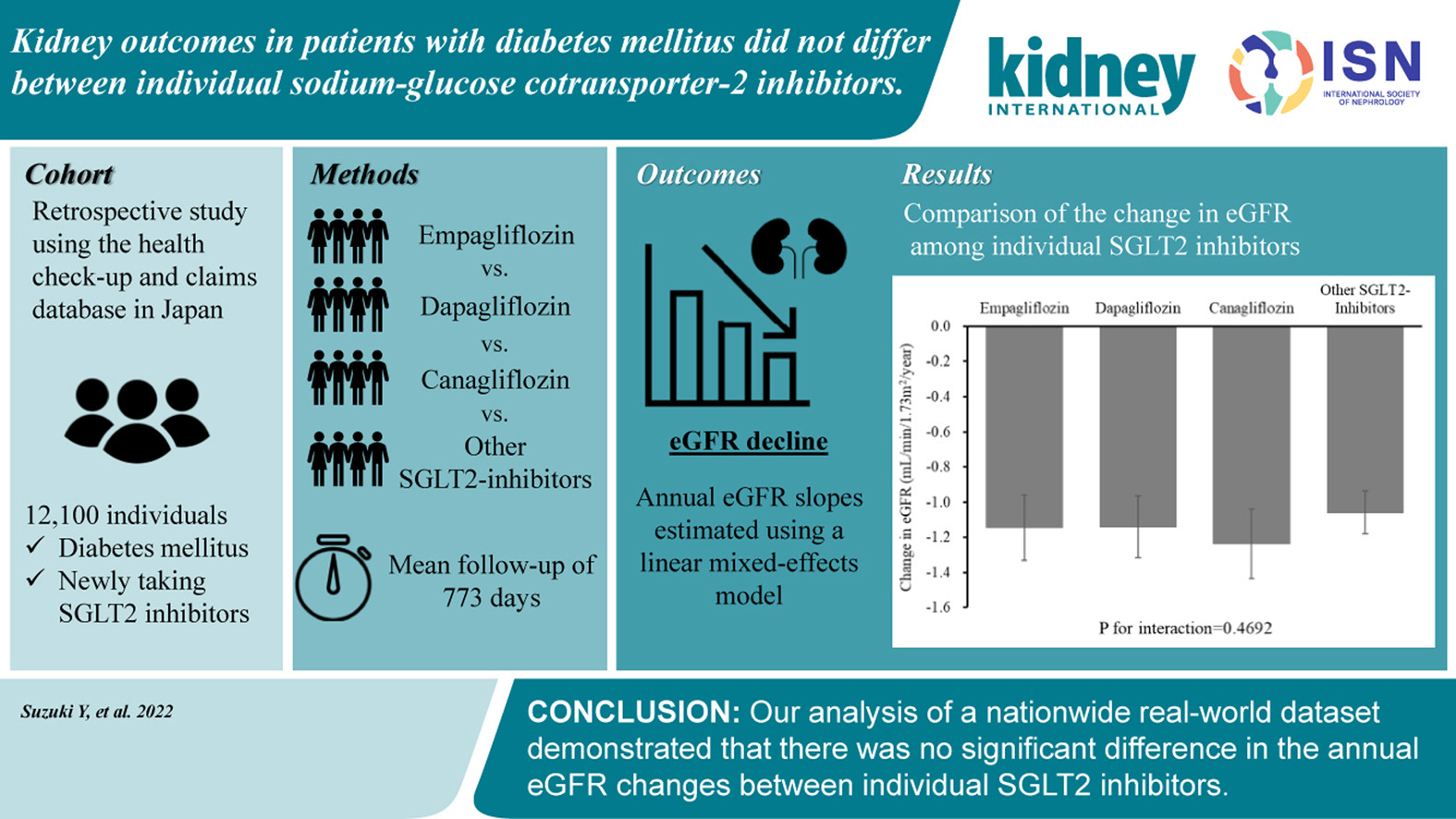
Due to their cardiovascular and renal benefits, SGLT2 inhibitors are now the first-line treatment for diabetes in people with chronic kidney disease (CKD). Several inhibitors are available for this group, with little comparative data available. This study compared the effects of initiating different SGLT2 inhibitors in diabetic patients with CKD using a large, real-world data set.
The authors analyzed data from over 20,000 patients with diabetes and CKD taking various SGLT2 inhibitors, including canagliflozin, dapagliflozin and empagliflozin.
The median baseline estimated glomerular filtration rate (eGFR) was 78 mL/min/1.73 m2 (interquartile range 68–90), and the mean follow-up period was 773 days. The primary outcome was the rate of estimated eGFR decline. The annual eGFR slopes of empagliflozin, dapagliflozin, canagliflozin, and other SGLT2 inhibitors were -1.15 (95% confidence interval, -1.33 to -0.96), -1.14 (-1.32 to -0.96), -1.24 (-1.44 to -1.04), and -1.06 (-1.18 to -0.94) ml/min/1.73 m2, respectively. No significant heterogeneity was noted among the different types of SGLT2 inhibitors.
Diabetes Management in Chronic Kidney Disease: A Consensus Report by the American Diabetes Association (ADA) and Kidney Disease: Improving Global Outcomes (KDIGO)
This report identifies and highlights shared recommendations from the ADA 2022 Standards of Medical Care in Diabetes and the KDIGO 2022 Clinical Practice Guideline for Diabetes Management in Chronic Kidney Disease and provides consensus guidance for optimal care.
The guidance addresses screening and diagnosis, comprehensive care, and a pharmacological approach to risk factor management with particular attention to glycaemic control, management of hypertension, dyslipidemia, and proteinuria with specific targets. Advice on special groups such as kidney transplant recipients and patients with estimated glomerular filtration rate (eGFR) <30ml/min is included.
Recommendations on using newer agents such as sodium-glucose transporter 2 inhibitors (SGLT2i), non-steroidal mineralocorticoid antagonists (ns-MRA), and glucagon-like peptide-1 receptor antagonists are made based on the latest evidence.
These include recommendations for:
- SGLT2i in most patients with type 2 diabetes (T2D ) and chronic kidney disease with eGFR >20 ml/min/ independent of glycated hemoglobin or the need for additional glucose lowering
- The addition of a GLP-1 receptor agonist with proven cardiovascular benefit for patients who do not meet their individualized glycaemic target with metformin and/or an SGLT2i
An ns-MRA with proven kidney and cardiovascular benefit (e.g., fineronone) is recommended for patients with T2D, eGFR >25 ml/min/ 1.73 m2, normal serum potassium concentration, and albuminuria (ACR >30 mg/g) despite maximum tolerated dose of RAS inhibitor, which remains the first-line treatment. The article includes tables and charts to aid decision-making.
Executive Summary of the KDIGO 2022 Clinical Practice Guideline for Diabetes Management in Chronic Kidney Disease: An Update Based on Rapidly Emerging New Evidence
The recent advent of the revised “KDIGO 2022 Clinical Practice Guideline for Diabetes Management in Chronic Kidney Disease” (CKD) has fast-tracked changes in practice thanks to landmark evidence-based trials.
This is a revolutionary era for everyone caring for diabetic patients with CKD. The updated guideline offers a well-defined approach to treatment, with an emphasis on individualized care for patients.
The 2022 updated guideline mentions a few specific changes from the 2020 edition, which mainly includes modifications on the use of SGLT2 inhibitors, GLP-1 receptor agonists, and the addition of a section on mineralocorticoid receptor antagonists. For increased tolerance to combination therapy, the sequential addition of medications affecting intrarenal hemodynamics with regular risk factor reassessment is necessary.
Maintaining improved lifestyle practices is a key factor in preserving kidney health in patients with diabetes. However, it is important to be mindful of
The beneficial role of medications for diabetic patients with CKD
The GFR cut-offs for their initiation
The timing for continuing and discontinuing these agents
For instance, an SGLT2i is to be initiated if GFR ≥20 ml/min per 1.73 m2 in the 2022 guideline compared to ≥30 ml/min per 1.73 m2 in the 2020 guideline. Once initiated, for maximum benefit, it should not be discontinued until initiation of renal replacement therapy.
SGLT2 inhibitor use is strongly recommended to prevent CKD progression and cardiovascular events, regardless of glycemia. Some of these agents form part of a ‘sick day’ protocol of which patients should be aware. GLP-1 receptor agonist use is preferred in patients with obesity, type 2 diabetes mellitus, and CKD; however, its use for weight loss is causing an increased global demand for this agent.
Although practice-changing research is ongoing, the last few years have been exemplary for the renal community with the addition of a few landmark trials that have proved to be game-changers, encouraging all involved in the care of people with diabetes and CKD and providing confidence in improved care for this group.
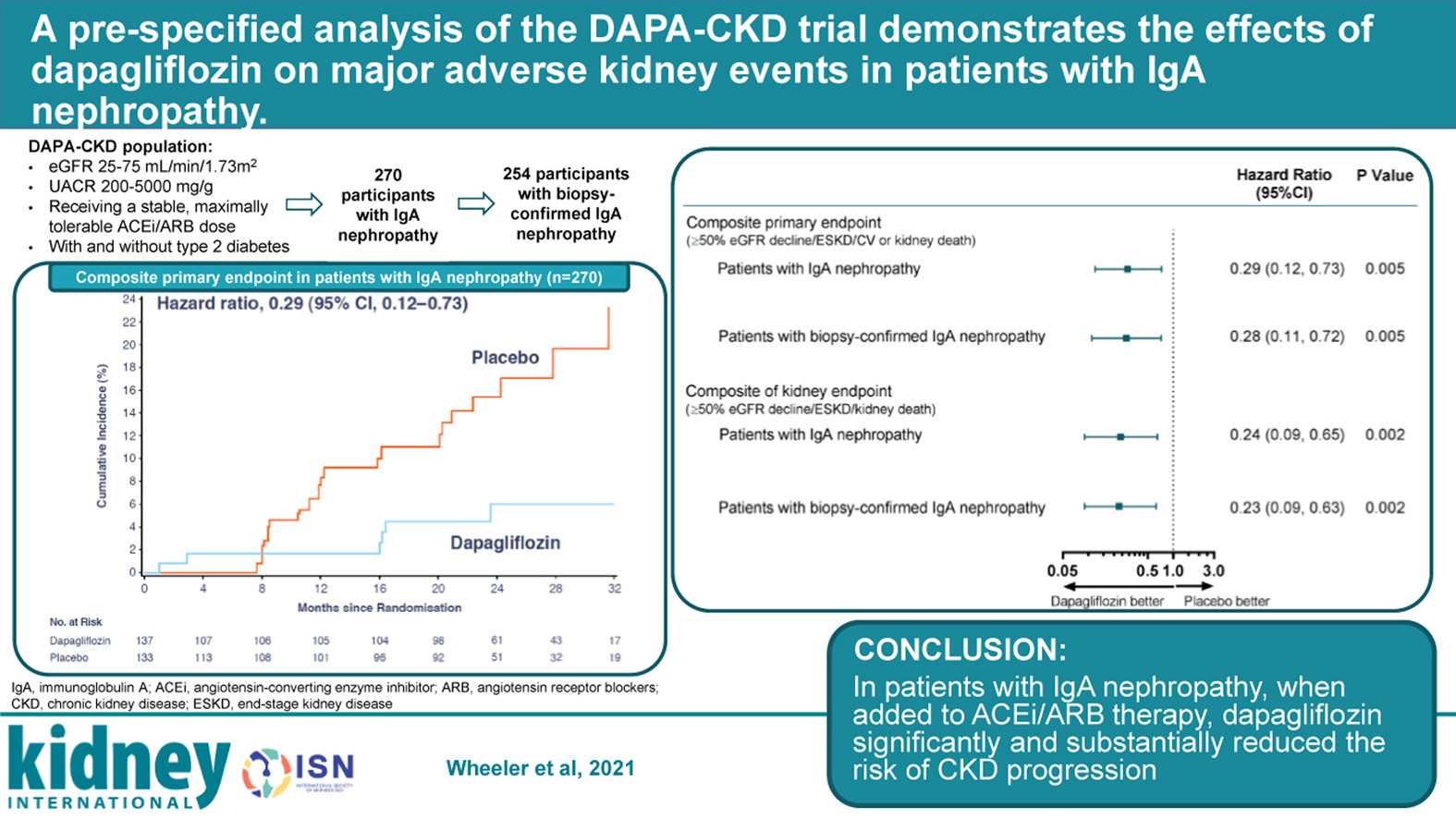
A Pre-Specified Analysis of the DAPA-CKD Trial Demonstrates the Effects of Dapagliflozin on Major Adverse Kidney Events in Patients With IgA Nephropathy
 There was a time when landmark trials for kidney diseases were unusual. However, with the advent of recent ground-breaking trials, the kidney care community can now confidently approach certain pathologies.
There was a time when landmark trials for kidney diseases were unusual. However, with the advent of recent ground-breaking trials, the kidney care community can now confidently approach certain pathologies.
MACE (major adverse cardiac events) was a common acronym, and now MAKE (major adverse kidney events) may also gain popularity.
Despite the use of RAS blockade and immunosuppressants, IgA nephropathy (IgAN) often progresses to kidney failure. Studies like TESTING and STOP-IgAN lack conclusive evidence that immunosuppression efficiently halts chronic kidney disease (CKD) progression in IgAN while proving their deleterious infection profile.
Dapagliflozin showed favorable outcomes in the DAPA-CKD trial with a reduction in kidney failure risk, decreased CKD progression, reduced urine albumin-creatinine ratio, and prolonged survival, proving to be a relatively benign yet effective addition to current practice in IgAN.
Although these outcomes are positive, another approach with budesonide use in patients already receiving ACEi/ARBs is being studied (NEFIGAN study). Also, the EMPA-KIDNEY trial is ongoing and includes a larger CKD cohort, hopefully strengthening findings of DAPA-CKD in IgA nephropathy.
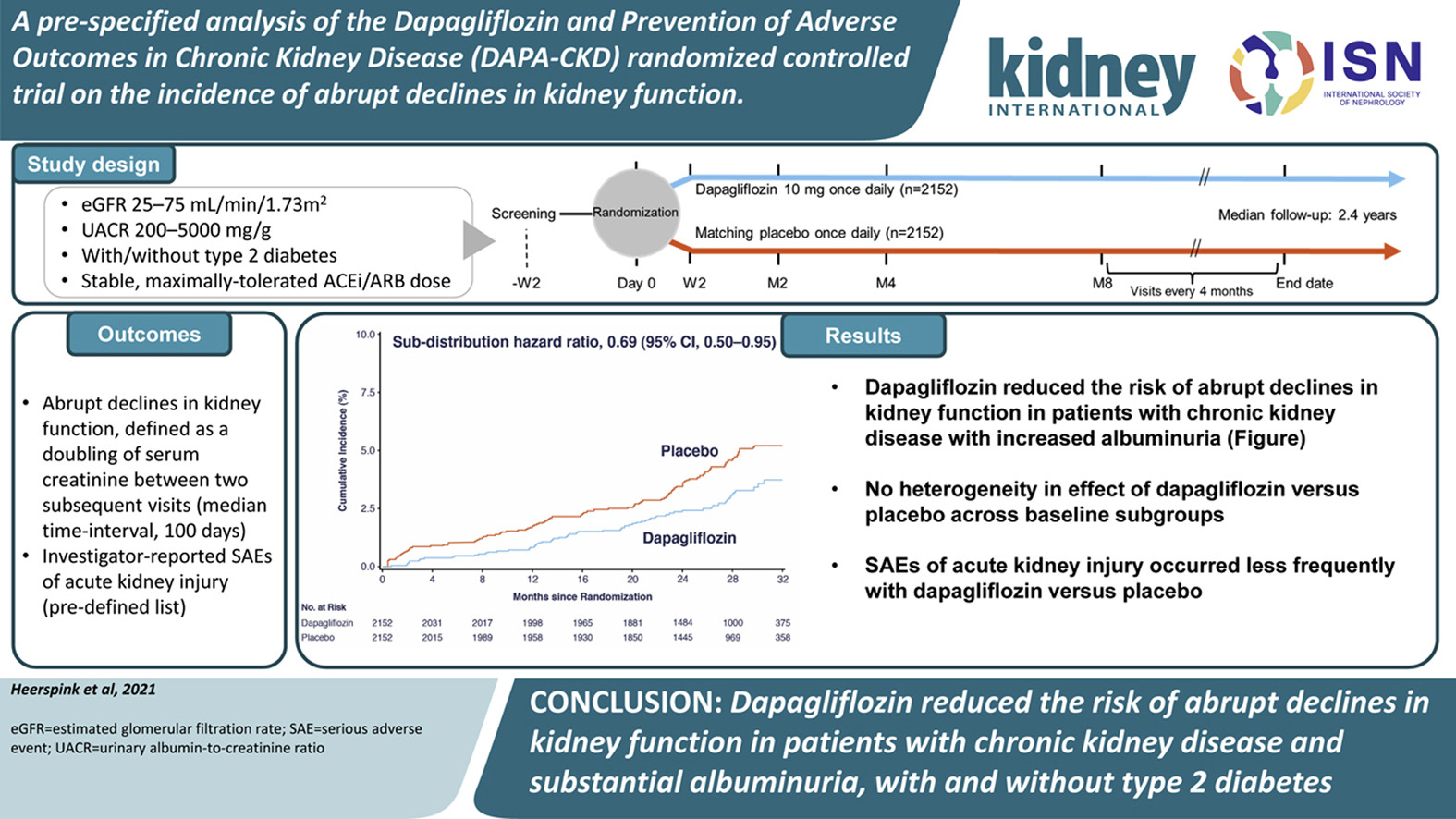
A Pre-specified Analysis of the Dapagliflozin and Prevention of Adverse Outcomes in Chronic Kidney Disease (DAPA-CKD) Randomized Controlled Trial on the Incidence of Abrupt Declines in Kidney Function
Acute kidney injury (AKI) in those with underlying chronic kidney disease (CKD) remains an important source of decline in renal function. AKI is also associated with adverse clinical outcomes, especially in patients with diabetes and those with significant albuminuria.
Sodium-glucose co-transporter 2 (SGLT2) inhibitors slow the decline in kidney function. Understanding the relationship between SGLT2 inhibition and the risk of AKI in patients with CKD and albuminuria is important, as patients with CKD experience higher rates of AKI than patients with normal or near-normal kidney function.
The DAPA-CKD trial demonstrated that dapagliflozin reduces the risk of kidney and heart failure hospitalization, and prolongs survival in patients with CKD, with and without type 2 diabetes.
This paper by Heerspink and colleagues looks at a pre-specified analysis of DAPA-CKD. It assesses the impact of SGLT2 inhibitors on abrupt declines in kidney function in high-risk patients based on having CKD and substantial albuminuria.
The authors found that AKI-related serious adverse events were not significantly different between the treatment and placebo groups and concluded that, if anything, dapagliflozin reduces the risk of abrupt declines in kidney function in people with CKD and substantial albuminuria.
The Impact of Dietary Nutrient Intake on Gut Microbiota in the Progression and Complications of Chronic Kidney Disease
The human gastrointestinal tract harbors a complex and dynamic population of microorganisms – the gut microbiota, which exerts a marked influence on the host during homeostasis and disease. The microbiota offers many benefits to the host through various physiological functions such as strengthening gut integrity or shaping the intestinal epithelium, harvesting energy, protecting against pathogens, and regulating host immunity.
Chronic kidney disease is associated with changes in the function and composition of the gut microbiota. This review by Koppe et al. explores our understanding of the diet-microbiota crosstalk in the context of uremia and how this may underwrite chronic kidney disease progression and complications.
This is followed by a discussion on how this knowledge can be used to develop personalized nutrition strategies to stop patients with chronic kidney disease from progressing to kidney failure.
KIDNEY INTERNATIONAL REPORTS ARTICLE |
Diabetes and chronic kidney disease (CKD) are linked to an increased risk of cardiovascular disease, which can lead to heart failure (HF) and death.
CKD is associated with changes in gluconeogenesis, an increase in glucose reabsorption, and insulin resistance, exacerbated by hypertension, obesity, chronic inflammation, and oxidative stress.
A hemoglobin A1c (HbA1c) goal of around 7% may be appropriate in the early stages of CKD. In advanced CKD, it should be personalized based on comorbidities and life expectancy.
Diabetes management in advanced CKD necessitates special considerations due to changes in glucose and insulin homeostasis, plus altered glucose-lowering therapy metabolism. Renal replacement therapy, such as dialysis, complicates management by limiting therapeutic options and predisposing individuals to hypoglycemia and hyperglycemia attacks.
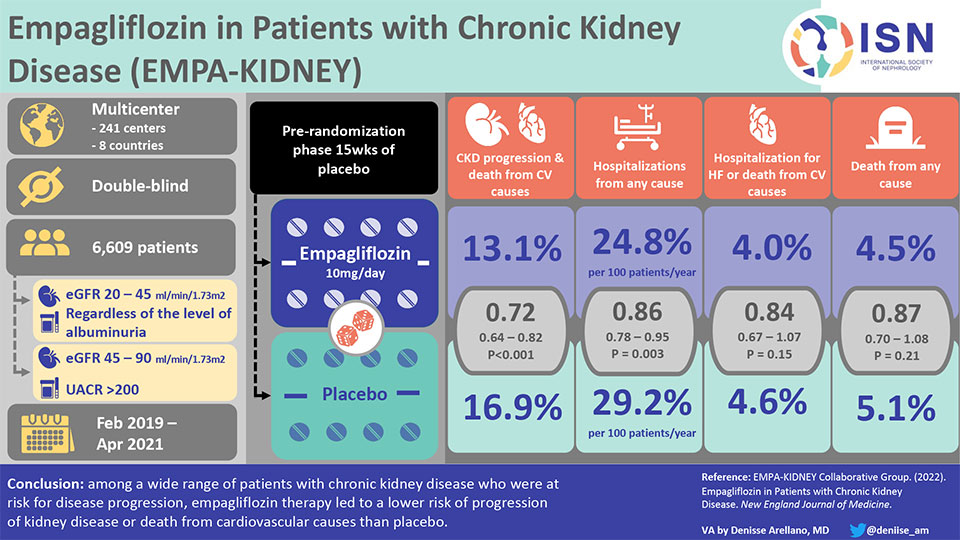
According to the results of the most recent clinical trials, including CREDENCE, DAPA-CKD, and EMPA-KIDNEY, sodium-glucose cotransporter 2 (SGLT2) inhibitors and their combination with glucagon-like peptide-1 receptor agonists (GLP1RAs) improve kidney and heart outcomes in patients with diabetes and CKD, regardless of HbA1c targets.
The combination of SGLT2 and SGLT1 inhibitors reduces composite cardiovascular death and hospitalization in the SCORED clinical trial. SGLT2 inhibitors are now part of standard medical therapy In this high-risk population.
Collaboration across healthcare specialties and disciplines will be required to provide optimal care for patients with diabetes and CKD.
In diabetic patients, SGLT2 inhibitors have emerged as an important disease-modifying therapy. Clinical trial evidence has expanded the indications for using these agents to prevent the progression of chronic kidney disease (CKD) and heart failure in diabetic and non-diabetic patients via mechanisms other than glycemic control.
The authors of this article focus on the practical considerations for prescribing SGLT2 inhibitors in stage 4 CKD patients with and without proteinuria, and heart failure patients. These include the initial acute decline in estimated glomerular filtration rate, diuretic effect, volume status, and mitigating adverse events.
The authors suggest considering glucagon-like peptide-1 receptor agonists and mineralocorticoid and endothelin receptor antagonists in combination with SGLT2I to reduce residual albuminuria and cardiovascular risks.
SGLT2 inhibitors have demonstrated that besides lowering sugar, they can improve kidney outcomes in people with chronic kidney disease independent of the presence of diabetes.
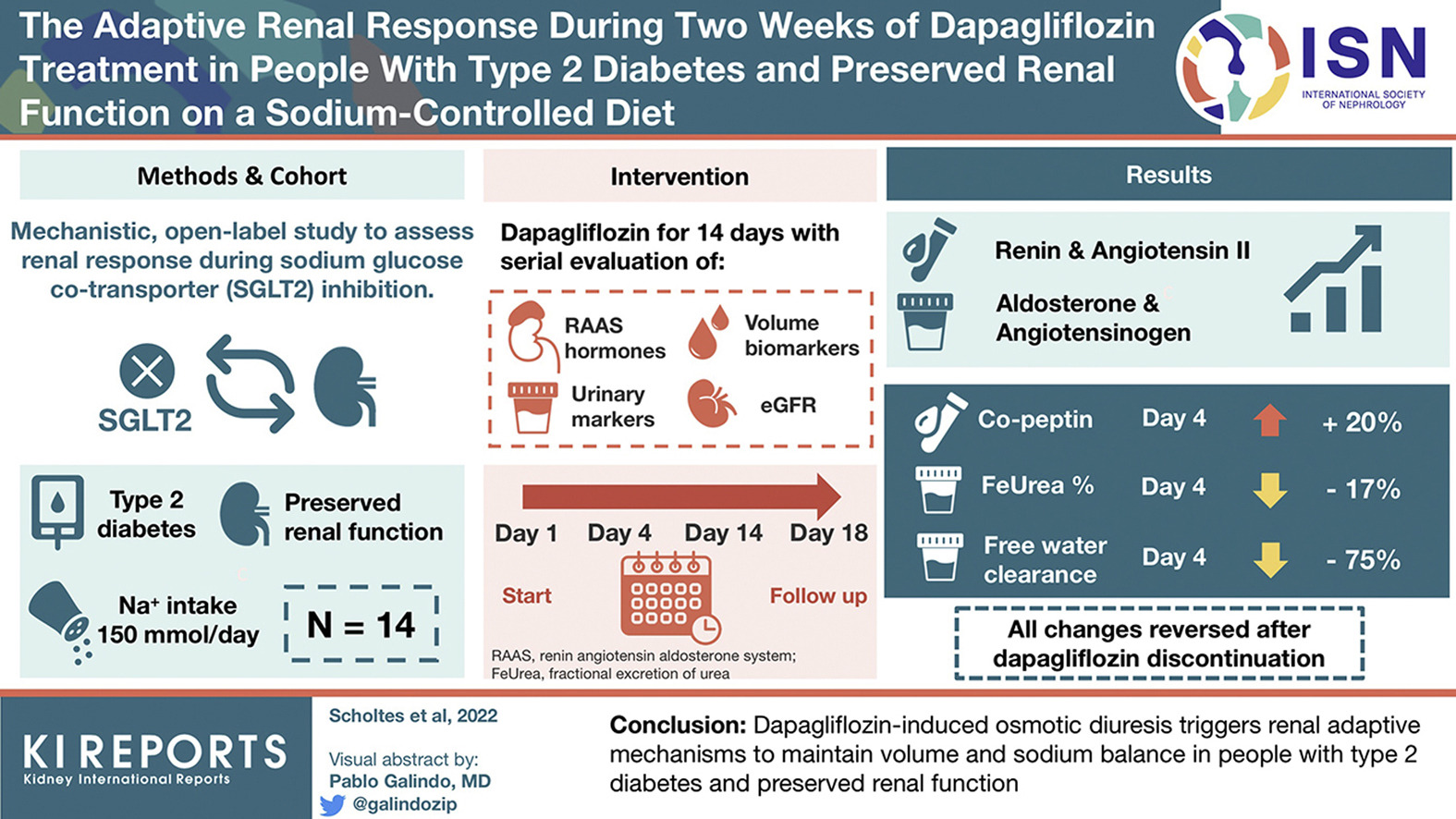
During SGLT2i treatment, adaptive changes occur to prevent excessive fluid loss. In this study, Scholtes et al. evaluated the effects of dapagliflozin on renin-angiotensin-aldosterone system hormones, volume-related biomarkers, urinary albumin-to-creatinine ratio and estimated glomerular filtration rate.
The authors found that patients on dapagliflozin presented increased levels of plasma renin and angiotensin and urinary aldosterone and angiotensinogen, which remained elevated during the treatment. Levels of copeptin increased after four days. Fractional urea excretion and free water clearance decreased significantly. After dapagliflozin was discontinued, the changes were reversed.
The DAPA-CKD trial showed that patients on SGLT2i presented reduced kidney and cardiovascular events and prolonged survival compared to placebo. This benefit was also observed in patients without diabetes.
The patients enrolled in the study belonged to four broad global regions: Asia, Europe, Latin America, and North America. Vart et al. found no differences in the efficacy and safety of dapagliflozin across geographic regions.
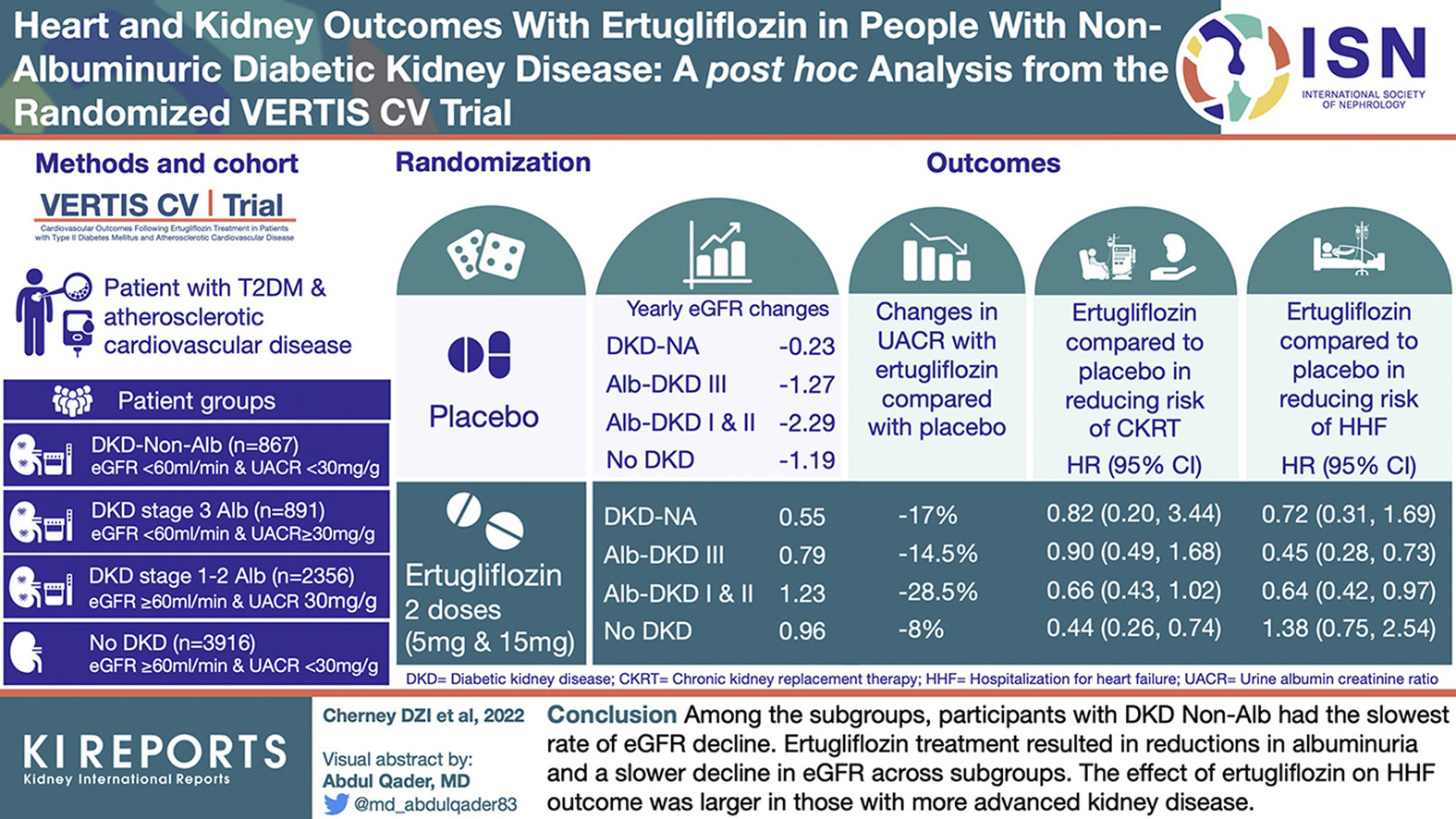
VERTIS CV was a cardiovascular outcomes trial that looked at the effects of ertugliflozin versus placebo in patients with type 2 diabetes with atherosclerotic cardiovascular disease. This trial used data from the VERTIS CV trial to demonstrate that ertugliflozin was non-inferior to placebo for major adverse cardiovascular events.
Cherney et al. looked at the impact of ertugliflozin vs. placebo in a subgroup of patients with non-albuminuric diabetic kidney disease. Subgroups were defined by estimated glomerular filtration rate (eGFR) and urinary albumin-to-creatinine ratios (UACR). The subgroups were defined: DKD-Non-Alb (eGFR < 60 + UACR < 30, n = 867); Alb DKD stage 3 (DKD stage 3 Alb, eGFR < 60 + UACR ≥ 30, n = 891); Alb DKD stages 1 + 2 (DKD stages 1–2 Alb, eGFR ≥ 60 + UACR ≥ 30, n = 2356); and no DKD (non-DKD, eGFR ≥ 60 + UACR < 30, n = 3916).
Ertugliflozin treatment resulted in a reduction in albuminuria and a slower decline in eGFR across all the subgroups. However, those in the DKD-Non-Alb group had the slowest rate of eGFR decline. Additionally, the effect of ertugliflozin on the hospitalization for heart failure group was greater in those with more advanced kidney disease.