April 2021 Edition

Summary Highlights
~ Vivekanand Jha
The COVID-19 pandemic continues to wreak havoc in many parts of the world. While some countries seem to have this in their rear view mirror, others are still struggling to manage the repeated surges.
The unique predicament of patients with kidney disease during the pandemic, especially those on dialysis and kidney transplant recipients, is well known to the global kidney health community.
The key strategy to overcome the pandemic is vaccination. The quick emergence of the large number of very effective vaccines has been a major triumph of the scientific community. Once the initial clinical trial results were out, the focus quickly shifted to managing the expansion of vaccination programmes in different countries.
Recognising the vulnerability of dialysis patients, a number of professional organisations led by The International Society of Nephrology called for prioritisation of this population for vaccination. Several countries, notably the United Kingdom, took heed of the compelling data generated by researchers and implemented this policy.
Patients with kidney disease were not included in most of the vaccine trials. Therefore, the global nephrology community must also study the effectiveness and safety of the vaccine in this population.
Such studies are ongoing, and results of a few of those have been presented in a collection in Kidney International.
These reports explore the immunogenicity of the currently available vaccines against SARS -CoV2 infection amongst patients on dialysis and amongst kidney transplant recipients.
KIDNEY INTERNATIONAL ARTICLES |
Immune Response to SARS-CoV2 Infection and Vaccination in Patients Receiving Kidney Replacement Therapy
COVID-19 vaccination efforts are underway worldwide. While the nephrology community has called for vaccination prioritization with ESKD patients, concerns exist that these patients may not mount a good response to vaccination. This commentary summarizes initial immunogenicity experiences from nephrologists around the world.
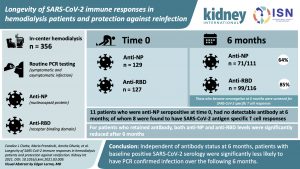
Longevity of SARS-CoV-2 immune responses in hemodialysis patients and protection against reinfection
Patients with end-stage kidney disease receiving in-center hemodialysis (ICHD) have had high rates of SARS-CoV-2 infection. The authors investigated the durability and functionality of immune responses to SARS-CoV-2 in these patients.
The results demonstrate a durable immune response six months post SARS-CoV-2 infection in patients receiving ICHD, with fewer than 3% of patients showing no evidence of humoral or cellular immunity.
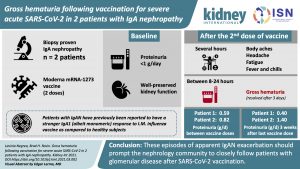
Gross hematuria following vaccination for severe acute respiratory syndrome coronavirus 2 in 2 patients with IgA nephropathy
Following episodes of apparent IgAN exacerbation after vaccinations using a nucleoside-modified, purified mRNA lipid nanoparticle-encapsulated platform, the authors encourage nephrologists to closely observe patients with glomerular disease after SAR2-CoV-2 vaccination to determine the frequency and consequences of vaccine-induced disease activation.
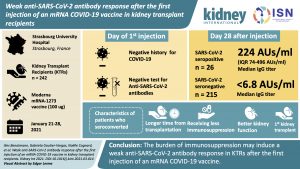
Weak anti–SARS-CoV-2 antibody response after the first injection of an mRNA COVID-19 vaccine in kidney transplant recipients
This study investigates the efficacy and safety of an mRNA COVID-19 vaccine in kidney transplant recipients (KTRs) by observing anti-SARS-CoV-2 antibody responses after the first injection.
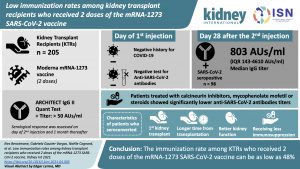
Results indicate that immunosuppression may induce a weak anti–SARS-CoV-2 antibody response in KTRs after the first injection of an mRNA COVID-19 vaccine compared with immunocompetent subjects.
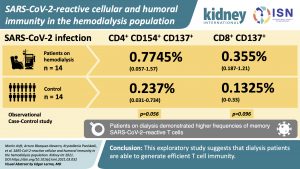
SARS-CoV-2-reactive Cellular and Humoral Immunity in Hemodialysis Population
This article describes an exploratory study which compares for the first time the magnitude and functionality of SARS-CoV-2 reactive T cells between dialysis patients and patients with normal renal function.
Low Immunization Rates Among Kidney Transplant Recipients who Received Two Doses of the mRNA-1273 SARS-CoV-2 Vaccine
Information on COVID-19 vaccine efficacy in kidney transplant recipients (KTRs) has been limited during this pandemic. This article reports immunisation rates of KTRs after having received two doses of the MRNS-1273 (Moderna) vaccine.
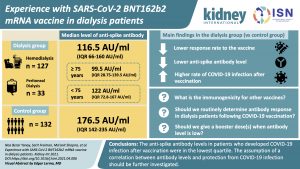
Experience with SARS-COV-2 BNT162b2 mRNA Vaccine in Dialysis Patients
Representation of chronic kidney disease (CKD) and end-stage kidney disease (ESKD) patients in initial clinical trials of COVID-19 vaccines has been low or unreported. This article investigates dialysis patients in Israel that had completed 2 doses vaccination of BNT162b2 and follows them for up to 10 weeks.
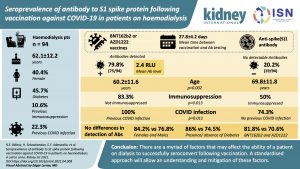
Seroprevalence of Antibody to S1 Spike Protein Following Vaccination Against COVID-19 in Patients on Haemodialysis.
A Call to Arms
Understanding the immune responses of patients on hemodialysis is vital to guide current and future vaccine dosing strategies in this vulnerable group. This article describes the antibody response 28 days after the first dose of either BNT162b2 (Pfizer-BioNTech) or AZD1222 (Oxford-AstraZeneca) vaccines in a diverse group of 94 patients on Maintenance Hemodialysis (MHD) in the United Kingdom.
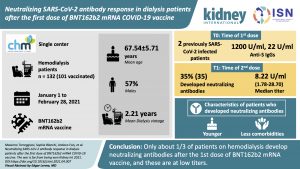
Neutralizing SARS-CoV-2 Antibody Response in Dialysis Patients after the First Dose of the BNT162b2 mRNA Covid-19 Vaccine. The War is Far from Being Won
On December 21, 2020, the European Commission granted conditional marketing approval to the BNT162b2 COVID-19 mRNA vaccine developed by BioNTech. This article provides a first report on the initial response to Covid-19 vaccine in patients on hemodialysis in France.
Antibody Response to BNT162b2 Vaccine in Maintenance Hemodialysis Patients
This article analyses the efficacy of the messenger RNA (mRNA) vaccine BNT162b2 in preventing COVID-19 in Maintenance Hemodialysis (MHD) patients, a group who are known to have altered vaccine responses.
KIDNEY INTERNATIONAL REPORTS ARTICLE |
Timing of COVID-19 Vaccine in the Setting of Anti-CD20 Therapy: A Primer for Nephrologists
This primer provides a review of literature on established vaccine use in anti-CD20 treatment and recommendations for both the timing of COVID-19 vaccines and measuring responses to administration. It serves as a guide to the pathobiology of B-cell–depleting therapy and as a useful aid to practitioners and trainees in nephrology.
All Visual Abstracts by Edgar Lerma (Member, @ISNEducation) with support from individual authors