June 2022 Edition

Impact Factor 2021
Articles of special interest to nephrologist in the field of acute kidney injury have recently been published in Kidney International. This eDigest highlights original papers in basic research and clinical investigation, including a ‘Special Report’ and a newly-adopted article type – ‘Next Generation Clinicopathological Conference’.
KIDNEY INTERNATIONAL ARTICLES |
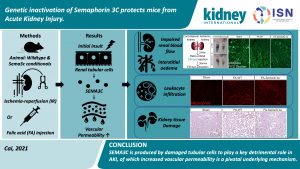
Genetic Inactivation of Semaphorin 3C Protects Mice from Acute Kidney Injury
SEMA3C is present in embryonic kidneys and disappears in adulthood. Its upregulation is specifically noted in diseased kidneys in both mice and humans, where kidney biopsies have evidence of tubular immunostaining for SEMA3C. Destructive mechanisms of acute kidney injury (AKI) are induced by SEMA3C causing vessel hyperpermeability, leading to impaired blood flow, interstitial edema, and inflammatory cell infiltration into renal tissue. Furthermore, rises in urinary SEMA3C levels were noted even before elevation in urea levels. Significant improvement in microvascular blood flow, along with a reduction in inflammatory cell infiltration into renal tissue, was seen in the group with downregulation of SEMA3C compared to the control group. Also, SEMA3C’s interaction with plexin-D inhibits lymphangiogenesis and its downregulation could alleviate renal damage by increasing lymphangiogenesis.
Anti-SEMA3A antibody has been used for sepsis in mice with positive results, and downregulation of SEMA3C alleviated AKI in mice models.
This knowledge makes this area of research exciting for advancement in therapeutic practice in nephrology. This data is beneficial for strengthening the understanding of AKI pathophysiology, investigating SEMA3C as an early indicator of AKI and its progression to CKD, and introducing novel targeted diagnostic and therapeutic interventions. However, further research is required in this field, with a need for human translational studies to investigate SEMA3C activity and its significance in AKI with the prospect of venturing into possible therapeutic mechanisms by finding inhibitors of SEMA3C.
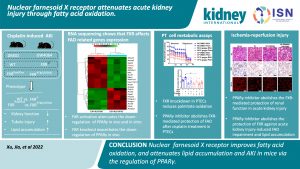
Nuclear Farnesoid X Receptor Attenuates Acute Kidney Injury Through Fatty Acid Oxidation.
Inhibition of fatty oxidation (FAO) leads to lipotoxicity and energy deprivation in renal proximal tubular cells during acute kidney injury (AKI). Farnesoid X receptor (FXR) is a transcription factor that regulates the expression of genes involved in FAO and peroxisome proliferator-activator receptor -g signaling. Activation of FXR has been shown to protect against AKI, but the mechanisms were unknown. The authors of this study, Xu et al., tested the hypothesis that increased FXR is protective in cisplatin-induced AKI (CP-AKI) through FAO and PPAR-g expression. This study provides insight into the mechanisms of AKI and suggests some potential strategies for therapeutic intervention.
The authors used a combination of in vivo and in vitro studies in mice to observe that cisplatin induces lipid deposition and reduces FAO in renal tubules by downregulation of FXR and FAO-related genes. This injury is exacerbated in FXR knockout mice compared to wild type and is attenuated by activation or over-expression of FXR. They further show that the adverse effects of FXR ablation are specific to the proximal tubule. PPAR-g upregulation was noted to be associated with FXR upregulation, PPAR-g agonists appeared protective against kidney injury and lipid accumulation, and PPAR-g antagonists and carnitine palmitoyl transferase-1 a inhibitors appeared to counter the protective effects of FXR overexpression. Finally, the protective effects of FXR/PPAR-g appeared to extend to ischemia/reperfusion models of AKI.
The authors concluded that the function of the FXR/PPAR-g axis is important in the development and prevention of AKI.
Kidney Repair and Regeneration: Perspectives of the Niddk (Re)Building a Kidney Consortium
Acute kidney injury and chronic kidney disease are common conditions associated with negative renal and patient outcomes. Unlike many other organs, the human kidney was not previously thought to have the ability to regenerate, having relatively low basal cellular recovering potential. More recently, this has been disputed, and it is now clear that the kidney has a real but limited regeneration ability. Understanding this process further may allow us to manipulate the course of renal injury and thus help develop therapeutic options to improve patient outcomes.
This paper by Naved and colleagues describes work from the (Re)Building a Kidney consortium (RBK), a National Institute of Diabetes and Digestive and Kidney Diseases undertaking tasked with finding ways to improve kidney repair, forestall progression to CKD, and devise innovative approaches to generation and engraftment of kidney replacement tissue. It further illustrates how different kidney cell types show varying potential for endogenous repair and tissue replacement. The publication presents multiple models of cellular injury and different approaches to accelerate the pace of discovery, thus allowing successful strategies to repair and regenerate injured kidneys.
Integrated Single Cell Sequencing and Histopathological Analyses Reveal Diverse Injury and Repair Responses in a Participant with Acute Kidney Injury: A Clinical-molecular-Pathologic Correlation
To define the cellular and molecular landscape, the authors used cutting-edge, single-cell technologies in conjunction with histopathology analysis of a kidney biopsy from a 30-year-old man in the transition stage between kidney injury and recovery time. The reported case is an example of how gene expression patterns at the single-cell level, when integrated with a robust, comprehensive atlas, can inform biological pathways that drive the shifts from healthy to injury state and from injury state to recovery in the individual patient. This new technique offers important possibilities for developing molecular algorithms that predict the outcome of AKI in a specific patient and identify pathways that can be targeted to avoid harmful consequences.
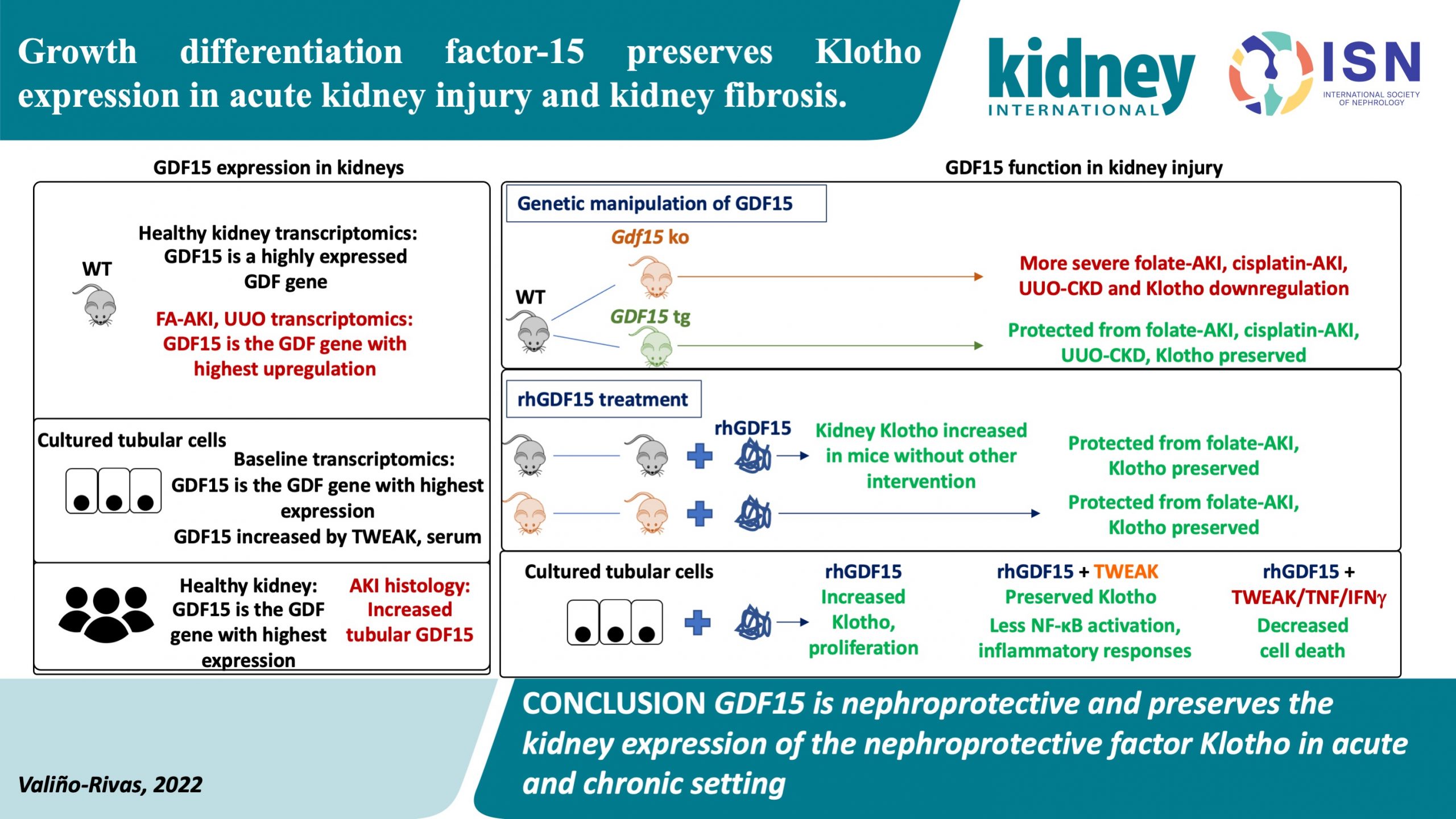
Growth Differentiation Factor-15 Preserves Klotho Expression in Acute Kidney Injury and Kidney Fibrosis
Anti-inflammatory, antiproliferative and antitumorigenic properties have been attributed to increased differentiation factor-15(GDF15). Elevated levels of GDF15 are found in humans with chronic kidney disease (CKD) and other chronic inflammatory diseases and are correlated with CKD progression and mortality. It is believed that GDF15 plays a deleterious role in kidney disease. Klotho is an anti-aging protein that plays a role in normal kidney physiology and has been used as a marker of kidney disease. Decreased levels of Klotho are reported in patients with acute kidney injury (AKI), kidney fibrosis and CKD. Its exogenous administration attenuates kidney disease and is considered a potential therapeutic target.
In this study, Valiño Rivas et al. explored the function of GDF15 in experimental AKI and kidney fibrosis and its relationship to the modulation of the expression of Klotho. They demonstrated a nephroprotective role of GDF15; in induced AKI, increased levels of GDF15 were found. In the absence of GDF15, the severity of AKI was higher, and the expression of Klotho was depressed. Exogenous administration of GDF15 provides kidney protection and preserves Klotho expression.
Harmonization of Epidemiology of Acute Kidney Injury and Acute Kidney Disease Produces Comparable Findings Across Four Geographic Populations
New approaches to study the health of the kidney allograft by non-invasive techniques also provide us with the means to understand the internal renal milieu and potentially intervene in a timely manner to prevent allograft dysfunction. Extracellular vesicles (EV), derived from the urine, provide a window into kidney donor quality and allograft rejection. In a timely review from Ashcroft et al, the formation of EV, their classification, isolation and characterisation are summarised, and their unique potential as novel therapeutic agents for prevention of ischaemia reperfusion injury and modulation of alloimmunity are discussed.
KIDNEY INTERNATIONAL REPORTS ARTICLE |
Recent publications in KI Reports investigate the relationship between biomarkers and kidney disease in various stages, ranging from AKI to CKD to kidney failure.
Considerations in Controlling for Urine Concentration for Biomarkers of Kidney Disease Progression After Acute Kidney Injury
Acute kidney injury (AKI) is a common complication in hospitalized patients, often secondary to ischemic or toxic injury. Many biomarkers are involved and have been evaluated for clinical application in AKI. Most of these biomarkers are measured from spot urine collection, and their values are often indexed to urea creatinine (UCr) concentration. However, there is no consensus on how urine concentration should be volume-controlled for AKI. Situations where UCr increases or decreases can alter biomarkers levels, leading to over or underestimation. Does this have a clinical impact?
In this cohort study, Wen et al. assessed the associations and predictions of urine biomarkers with incident chronic kidney disease (CKD) and CKD progression. They report that UCr and urine osmolality (UOsm) during hospitalization were inversely associated with the development of CKD. Also, they found that some biomarkers, such as KIM-1, IL-18 and MCP-1, are associated with CKD after the indexation for UCr or UOsm.
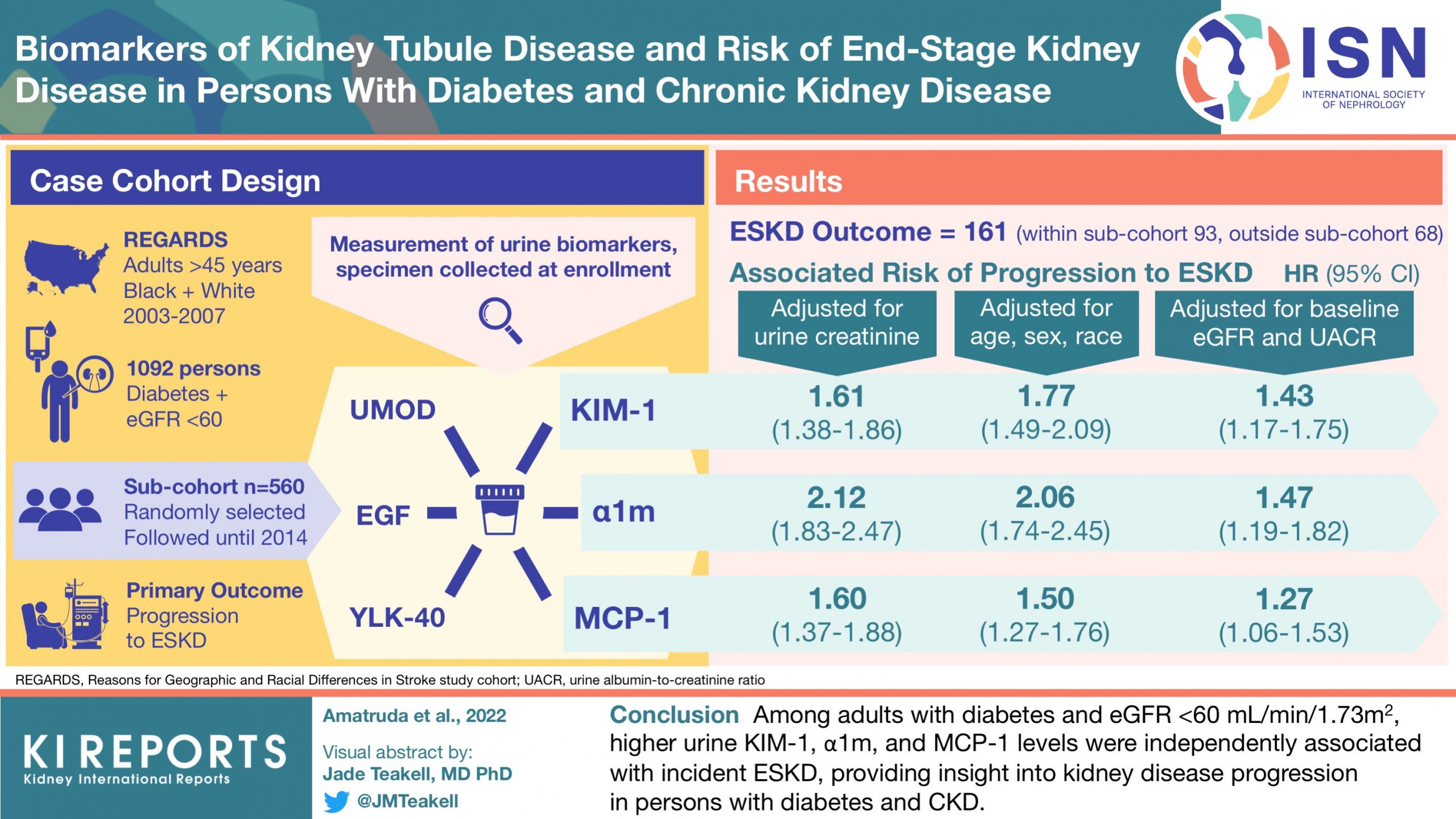
Biomarkers of Kidney Tubule Disease and Risk of End-stage Kidney Disease in Persons with Diabetes and Chronic Kidney Disease
Serum Creatinine and uACR are currently gold standards of kidney function assessment, but we rely heavily on biopsy to detect tubular insufficiency. Biomarker detection is emerging as an important indicator of tubular injury and dysfunction at a cellular level. In this study, every two-fold rise in urine KIM-1, α1m, and MCP-1, independent of eGFR and uACR, was associated with ESKD, supporting the hypothesis that tubular damage is an important pathway of CKD progression in diabetes.
There are certain challenges to overcome before establishing these biomarkers into broad clinical practice for detecting and prognosticating tubular disease in diabetics and CKD patients, and mitigating the risk for progression to ESKD. Combining these specific biomarkers into lab panel assays will be increasingly useful for global kidney health, especially where accessibility to kidney biopsy is limited.
Despite the accuracy of these tubule-specific biomarkers, further research is required, and biopsy will remain crucial to accurately detect tubular damage, especially because biopsy can reveal unexpected findings that may alter management.
Biomarkers will be essential in cases where biopsy is contraindicated, and accessibility is limited. Kidney biopsy also has limitations – providing only a snapshot picture at a single point in time – and does not help assess disease progression. Globally, immunofluorescence and electron microscopy availability is limited, and there is interobserver disagreement on the interpretation of pathology. If urinary biomarkers are used in conjunction with an initial biopsy, we can efficiently correlate disease progression and intervene with proper management without unnecessarily exposing the patient to repeated invasive biopsies for assessment.
Urinary Trace Elements are Biomarkers for Early Detection of Acute Kidney Injury
Current definitions of acute kidney injury (AKI) are based on increased serum creatinine or decreased urine output – markers of kidney filtration rather than early parenchymal injury. There is still no ideal biomarker for detecting early AKI. In this study, the authors explored the place of urinary trace elements (TE) as biomarkers for early detection of AKI.
Using a porcine model of ischemia-induced AKI they detected urinary cadmium (Cd), copper (Cu), and iron (Fe) as potential biomarkers. These were then tested in two observational cohorts of patients at risk of developing AKI, namely those undergoing cardiac surgery (n=151) and ICU admissions (n=150).
All TE increased after cardiac surgery and in ICU patients with AKI. Urinary Cu, Cd and Zn had negative predictive values for acute kidney injury with limited sensitivity. Urinary Fe was less clinically useful due to confounding variables. The biomarkers were stable at room temperature for 14 days and were not affected by the presence of chronic kidney disease.
Plasma Biomarkers as Risk Factors for Incident Chronic Kidney Disease
An estimated 850 million people globally have chronic kidney disease (CKD), with an antecedent projected growth that will impose a significant burden in the future. 90% of these patients are at early stages, and preventing further damage before reaching the point of irreversible fibrosis would be ideal. A major limiting factor is a lack of sensitive biomarkers to quantify risk, especially in non-diabetics. Serum creatinine remains the only biomarker used to estimate GFR, but it is relatively insensitive at the early stages of kidney damage. Sensitive early markers are needed to prevent CKD progression.
In this study, Sarnak and Colleagues in the US evaluated an association of novel plasma biomarkers with incident CKD using a case-cohort design in patients without diabetes with a baseline GFR of 60 or above. Incident CKD is defined as development of eGFR < 60 or a 40% decline from baseline. Six plasma biomarkers were evaluated, and Cox regression models used to estimate hazard ratios of incident CKD. The paper concludes that plasma concentrations of biomarkers TNFR-1 and TNFR-2 are consistently associated with incident CKD in non-diabetic patients. It is likely that if these biomarkers are adapted for early use and become accessible for everyday use, we may be able to slow down the deluge of CKD in the future.
A Prospective Evaluation of Novel Renal Biomarkers in Patients with Lymphoma Receiving High-Dose Methotrexate
High-risk lymphoma patients require high doses of methotrexate (MTX), and the efficacy and toxicity of MTX are dependent on rapid elimination by the kidneys. In this prospective study, serum creatinine as a surrogate marker is challenged by novel markers of acute kidney injury.
Urinary neutrophil gelatinase-associated lipocalin (NGAL) failed to demonstrate any utility in this cohort, whereas patients with urinary tissue inhibitor metalloproteinase-2 concentrations higher than baseline were significantly more likely to develop any AKI.